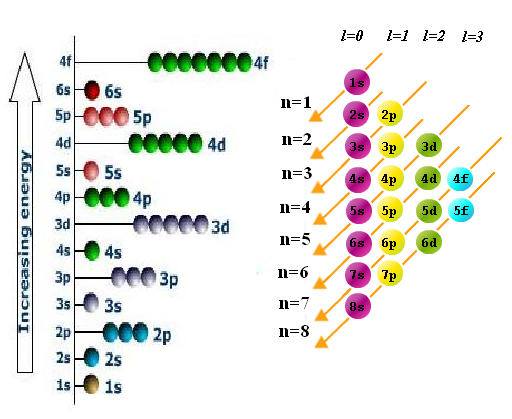 
The three orbitals in the 2 p subshell)? Orbitals of the same energy level are known as degenerate orbitals, and we fill them using Hund’s rule: place one electron into each degenerate orbital first, before pairing them in the same orbital. But how should we fill multiple orbitals of the same energy level within a subshell (eg. Generic energy diagram of orbitals in a multi-electron atom.Īs previously discussed, the Pauli exclusion principle states that we can only fill each orbital with a maximum of two electrons of opposite spin. This is demonstrated in Figure 1.1, where each line represents an orbital, and each set of lines at the same energy represents a subshell of orbitals.įigure 1.1. The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n. In many-electron atoms (all atoms except hydrogen), the energy levels of subshells varies due to electron-electron repulsions. Increasing the principle quantum number, n, increases orbital energy levels, as the electron density becomes more spread out away from the nucleus. The overall pattern of the electron shell filling order emerges from the Aufbau principle (German for “building up”): electrons fill the lowest energy orbitals first. We have just seen that electrons fill orbitals in shells and subshells in a regular pattern, but why does it follow this pattern? There are three principles which should be followed to properly fill electron orbital energy diagrams:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
